|
Back to Panama, for the fifth fieldwork season of data collection of my PhD. I have three determined goals for this session : (i) collecting survival data, (ii) collecting DNA samples and (iii) manipulating group size. This is the third consecutive season where I am catching a network of a dozen roosts where I can determine group size.
During captures, animals are marked individually with transponders. With relevant statistical analyses, I will try and estimate survival based on recapture rates and detection probabilities. My species foraging in groups, group size probably has an effect on individual survival, that’s what we are trying to determine with these analyses. In parallel, I am collecting wing biopsies. These DNA samples will be genotyped to obtain microsatellites. They will be used to study relatedness of individuals within groups. I am especially interested in male reproductive success in function to group size. I assume it will be more difficult for the dominant male to insure paternity in groups with more females. And the main goal of this mission is to study the foraging efficiency (weight gain relative to time spent out of roost) in function to group size. Several roosts studied are equipped with automated systems with automated transponder reader and integrated scale. To investigate the effect of group size, I am comparing foraging efficiency for a group of normal size and of modified size. To modify group size, I capture individuals of the group that I keep for several days in captivity. Captive individuals are fed each night with mealworms and released within few days. I have already manipulated three groups, a fourth is ongoing and a fifth will come in the days to come. I have now collected a lot of data, in parallel I am now working on the analyses.
0 Comments
This video has been made with a drone, owned by a colleague from the Smithsonian Tropical Research Institute, the institute with which I collaborate in Panama. This is basically an aerial discovery of my fieldwork site. My bat model species roost in the houses of this village - all the data from my PhD is coming from there...Enjoy! I stayed a short month in Panama this summer – from mid-July to mid-August. The priority was to catch all the colonies we work on – more than 20 nights of catching for a total of around 300 bats. Many new bats have been transpondered and I have collected a lot of DNA (relatedness analysis) and feces samples (virus and diet studies). The highlight of the stay was the catch of 96 bats in one night. The catch started at 6PM and we finished processing the bats around 6AM.I had a small army of people from the batlab to help me – thanks to Teague, Basti, Julia, Sebas, Michelle, Toni, Santi, Dallas…
Unfortunately, three of the roosts with automated monitoring systems were abandoned (probably due to house renovation) but we installed two other automated monitoring systems. I also had the opportunity to present my PhD project to the scientific community – good practice with a lot of interesting questions. In addition, I helped Sebas for a parallel project. As a pilot study for his master thesis, we equipped these bats with heart-rate and temperature transmitters. The bats were kept in captivity and video-taped. This preliminary data will help understanding the relationship between heart-rate, temperate and clustering prevalence. My fieldwork season is over but Sebas will stay until early September to operate more trials and continue this project in November-December, when we both go back for a new fieldwork season. I am in the starting-blocks for a new fieldwork season in Panama. I will fly to Panama mid-July and stay until mid-August. My goal for this season is to catch all the colonies already surveyed in the previous fieldwork seasons - especially to collect DNA samples and equip non-marked bat with a subcutaneous transponder. This transponder can be scanned either manually (hand-held transponder reader) or automatically (automated transponder reader at roost entrance). This method is very useful to obtain data on dispersal, group composition and also survival.
A german Master student will help me for the catching and removal of bats from the nets. In parallel, this assistant will conduct some captivity experiments for his master's thesis. His project will focus on heart-rate and temperature to better understand metabolic rates of tropical animals. This should be the last but one fieldwork season of my PhD, the last one being planned in November-December. I will then have one year and a half to go in the lab for my DNA samples, analyse the collected data and write my PhD thesis. We have been tracking two species of bats in the last weeks.
For Teague's project on information transfer, we tracked Uroderma bilobatum, a small leaf-nosed bat eating figs. During 10 days, we tracked a handle of bats from 7PM to 5AM to learn more about their foraging grounds and see if the bats were foraging together. Two of our bats seemed to forage at the same fig tree but we need additional studies to prove it. In parallel, we tracked a roost of 11 Molossus molossus - the model species of my PhD. The idea of this tracking session was to assess the prevalence of group hunting in this species. Unfortunately, all the collars with the transmitters were removed after 2 days. We moved on to additional tests of collars in captivity but we could not design a collar that would stay long enough on bats. The tracking session of Molossus molossus is cancelled, we are now focussing on data collection at roosts and also analysing data. I'm going back next week for a third fieldwork season in the village of Gamboa Panama. Program: to continue data collection on my doctoral model species: Molossus molossus. And a field mission requires a bit of organisation !
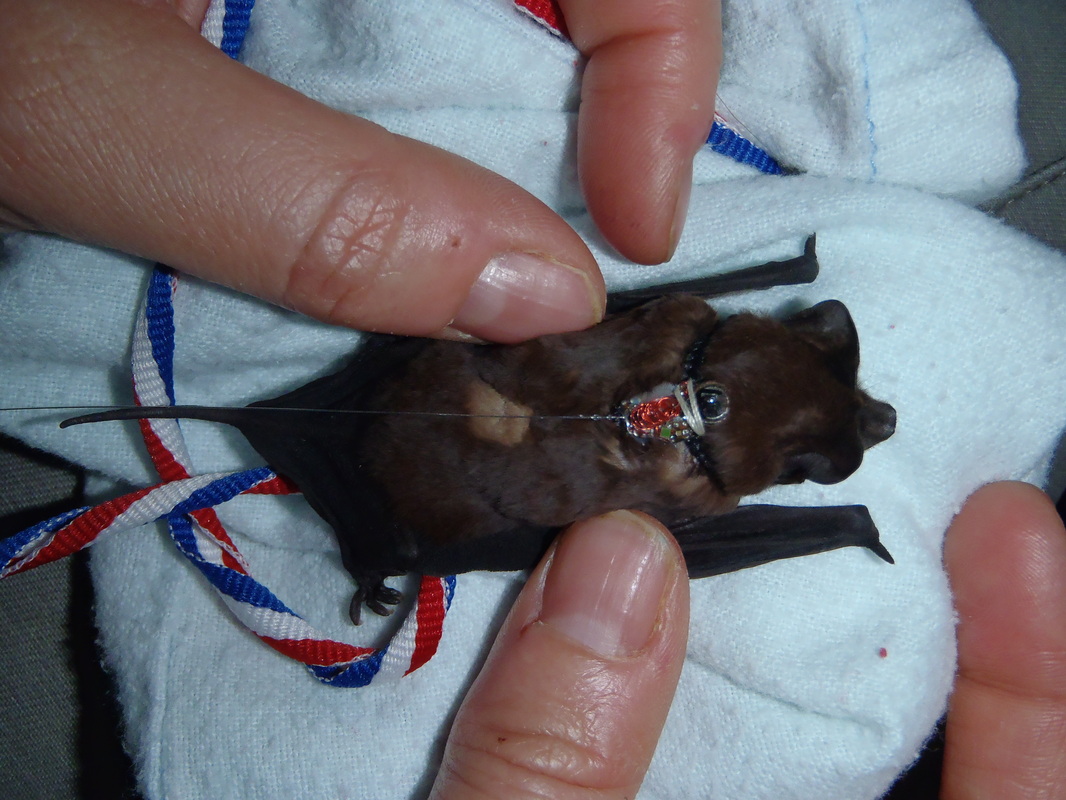
As with all trips, I organize for me and my assistant the housing and transportation (plane, train, taxi ...). Together with my french assistant, we will live in an apartment provided by the host institution, the Smithsonian Tropical Research Institute. These homes are located in the village, near my roosts of bats. To complete my research legally, research permits for sample collection (skin, guano) must be written in Spanish and sent to the Panamanian authorities before verification. When my fieldwork season comes to an end, I will send a report of my fieldwork season and ask for an export permit to bring back to Germany the samples collected. And to complete the organization for the field, preparation of material is essential. Checking nets, preparing telemetry equipment, electronic testing, preparation of materials for sample collection ... the list is long! And before I go, I am working on my fieldwork program to plan the 11 weeks in the field. Catch sampling, radio-tracking ... More news when I'm on the ground! For my Phd fieldwork in Panama, I use a wide variety of tools to measure, to transponder and to sample the bats I catch, often with help from fieldwork assistants or some colleagues. I use generally 7 tools, described here and shown on the photo below. 1. The first tool is a scale, used to measure the weight of the bat (placed in a soft clothed bag). This feature gives a good idea about the age of the bat or if it has eaten. For examplen, my species weighs around 10g and can come back from foraging weighing 3-4g more. 2. The second tool is a calliper, used to measure the forearm length. This feature is often used as an help for species determination. 3. The third and fourth tools are a transponder needle mounted on a transponder injector. The needle contains the transponder that will be quickly injected under the bat skin of the bat. This is a sensitive operation but I trained and I process quickly. The transponder - with an individual identification number - will be injected once and will last for the whole life of the bat. Once the transponder is inserted, I check the number with a hand transponder-reader. 4. To sample DNA, I use a biopsy wingpunch. This tool consists of a 3mm-diameter metal ring mounted on a plastic pole. The DNA is sampled on the wing membrane, the "wingpunch" is then stored in a small tube of ethanol. The sampling on the wing membrane is optimal because this body part contains a lot of DNA and the healing really quick (around 2 weeks). Another story starts after for exportation of the samples, extraction of the DNA and of course the analyses... Thanks to Hyuen-Ji Lee for the picture :).
PS: All catching and sampling procedures are realised with permissions from the Panamean authorities. A year has passed since I have started my PhD.
A lot of my time and energy went into this fieldwork season (mid-March until end July) to catch and transponder some bat colonies of my model species - Molossus molossus - in the village of Gamboa in Panama. After numerous hours of wood cutting and video check, I managed to instal automated balances that are monitoring bat activity thanks to their transponders. A contact in Gamboa is sending me the data by e-mail on a weekly basis. And my current program for the weeks to come is to try and extract the best information from this great source of data. I am also reading and writing to move forward on my PhD introduction – in parallel to fieldwork organization for the session of November-December (6 weeks) and the big tracking session in Spring… Stay connected! A little article on PIT tags was published in June's STRI newsletter. My boss and I use this technology
to recognize marked bats using a hand-transponder reader and also to recognize bats when they crawl through the automated balances that I have installed in Gamboa. "How does one track the comings and goings of bats, birds, frogs and other animals? A rice-grain sized PIT tag injected under the skin makes it possible to identify individuals using a hand-held tag reader. Passive Integrated Transponder tags consist of a small glass capsule containing electromagnetic coils that act as a combined receiver/transmitter. When triggered by an interrogating signal, the antenna in the tag uses voltage generated by the signal to radiate an alternating magnetic field coded with a unique ID number. This technology was first developed in the 1940’s to distinguish friendly from enemy aircraft. The same concept lets you open a door by waving a card in front of a reader. Since the 1980’s PIT tags have been employed to monitor fish populations and to identify valuable zoo animals and pets. Radio Frequency Identification Technology in printed tags tracks goods in the global supply chain. Seven millimeter-long PIT tags cost a little less than $5.00 each." |
AuthorLittle blog about my bat research and conservation action |